Understanding Tissue Trauma, How Computational Models Predict Injury Risk Using Matlab
Author : Waqas Javaid
Abstract
This study presents a computational biomechanical simulation designed to analyze human tissue response to dynamic impact and assess injury risk. Using a viscoelastic material model within a Hertzian contact framework, the simulation tracks key metrics including impact force, tissue stress and strain, and established injury criteria such as the Head Injury Criterion (HIC) and Viscous Criterion (VC) over a 0.1-second impact event. The model incorporates a logistic regression-based injury risk probability function to provide real-time severity classification [1]. Results demonstrate the critical relationship between strain rate, energy dissipation, and tissue failure thresholds, highlighting the importance of viscoelastic properties in mitigating injury [2]. This analytical framework offers a valuable tool for optimizing protective equipment design and advancing quantitative biomechanical safety assessment.
Introduction
Understanding the mechanical response of human tissue to dynamic impact is a fundamental challenge in injury prevention science. Each year, millions of individuals suffer traumatic injuries from falls, vehicular accidents, sports collisions, and occupational hazards, many of which result in permanent disability or death. Yet, directly testing human tissue response under injurious conditions remains ethically and practically impossible.

Figure 1 presents the computational biomechanical simulation of impact dynamics for injury prevention, incorporating stress–strain response analysis, Head Injury Criterion (HIC) and Viscous Criterion (VC) injury metrics, and energy dissipation modeling to evaluate human tissue response under impact loading conditions. This limitation has driven the development of computational biomechanics, where sophisticated mathematical models simulate the complex interactions between impact forces and biological materials. Human tissue exhibits unique mechanical behavior characterized by viscoelasticity, meaning its response depends not only on the magnitude of force but also on the rate at which that force is applied [3]. This time-dependent property makes injury prediction particularly challenging, as rapid loading can cause catastrophic failure even at relatively low force levels. Modern injury criteria, such as the Head Injury Criterion (HIC) and the Viscous Criterion (VC), have been developed to quantify this relationship and establish safety thresholds [4]. However, these metrics alone cannot capture the full complexity of tissue damage, which involves cumulative strain, energy dissipation, and material failure at the microstructural level [5]. This study presents a comprehensive computational framework that integrates viscoelastic material modeling, dynamic impact simulation, and multi-criteria injury assessment to predict tissue response and injury risk with high fidelity [6]. By simulating the complete mechanical event from initial contact through peak deformation and recovery, this approach provides a powerful tool for designing safer protective equipment and advancing our fundamental understanding of trauma biomechanics [7].
1.1 The Global Burden of Traumatic Injury
Traumatic injuries represent one of the most significant public health challenges worldwide, claiming millions of lives annually and leaving countless others with permanent disabilities. Motor vehicle collisions alone account for over 1.3 million deaths each year, while sports-related injuries and workplace accidents contribute substantially to this burden. Beyond the immediate human toll, these injuries impose staggering economic costs on healthcare systems and societies at large [8]. The financial impact of trauma care, rehabilitation, and lost productivity runs into hundreds of billions of dollars annually across developed nations. This profound societal challenge underscores the urgent need for more effective injury prevention strategies grounded in rigorous scientific understanding [9].
1.2 The Ethical and Practical Barriers to Human Tissue Testing
Despite the clear need for injury prevention research, direct experimental investigation of human tissue response to injurious forces remains fundamentally constrained by ethical considerations. Researchers cannot ethically subject human subjects to impacts that may cause serious injury or death, making traditional experimental approaches impossible [10]. Even cadaveric studies, while valuable, face significant limitations including tissue degradation, lack of physiological response, and ethical considerations surrounding donor consent. Animal models, though commonly employed, introduce interspecies variability that limits direct translation to human biomechanics. These constraints have created an urgent imperative for alternative approaches that can accurately predict human tissue behavior without exposing living subjects to harm [11].
1.3 The Emergence of Computational Biomechanics
In response to these experimental limitations, computational biomechanics has emerged as a transformative approach to injury prediction and prevention. This discipline combines principles from engineering mechanics, materials science, and biology to create mathematical models that simulate the behavior of biological tissues under mechanical loading. Advanced computational methods enable researchers to explore injury mechanisms in ways that would be impossible, unethical, or prohibitively expensive through physical experimentation alone [12]. These simulations can be repeated thousands of times with varying parameters, allowing for comprehensive sensitivity analysis and optimization of protective designs. The rapid advancement of computing power has made increasingly sophisticated simulations possible, bringing us closer to truly predictive models of human injury response [13].
1.4 Understanding Viscoelasticity in Biological Tissues
A fundamental challenge in modeling human tissue lies in its complex viscoelastic nature, which distinguishes it from simple engineering materials like metals or plastics. Unlike elastic materials that deform and return to shape instantaneously, or viscous fluids that flow continuously under stress, biological tissues exhibit behavior that combines both characteristics in a time-dependent manner [14]. When subjected to rapid loading, tissues behave stiffly and may fracture catastrophically, whereas the same tissue under slow loading may stretch considerably without damage. This rate-dependent behavior means that injury risk cannot be determined by force magnitude alone; the speed of loading is equally critical [15]. Any accurate injury prediction model must therefore incorporate viscoelastic material properties that capture this complex, time-dependent mechanical response.
1.5 The Role of Injury Criteria in Safety Assessment
To translate mechanical measurements into meaningful injury predictions, researchers have developed standardized injury criteria that correlate physical parameters with clinical outcomes. The Head Injury Criterion (HIC), developed through decades of automotive safety research, relates head acceleration to the probability of traumatic brain injury based on analysis of cadaveric and animal studies [16]. Similarly, the Viscous Criterion (VC) has emerged as a powerful predictor of soft tissue injuries by combining tissue deformation with the rate at which that deformation occurs. These criteria establish quantitative thresholds that distinguish between safe and injurious loading conditions, forming the backbone of modern safety standards [17]. However, each criterion captures only specific aspects of injury, necessitating multi-criteria approaches for comprehensive risk assessment.
1.6 The Complexity of Tissue Damage Mechanisms
Beyond simple force or acceleration thresholds, actual tissue injury involves complex mechanical and biological processes that unfold at multiple scales. At the macroscopic level, excessive stress and strain can cause immediate structural failure through tearing, fracturing, or rupture of tissues. At the cellular level, mechanical forces can trigger mechanotransduction pathways that initiate inflammatory responses, apoptosis, and other biological cascades that may lead to delayed injury manifestation. Repeated sub-threshold loading can accumulate damage over time through mechanisms such as fatigue, where individual impacts below the acute injury threshold collectively lead to tissue failure [18]. The interaction between these various damage mechanisms makes injury prediction inherently multifactorial, requiring models that can integrate multiple failure modes. A comprehensive simulation must therefore track not only peak values but also cumulative damage indicators that reflect the complex nature of biological tissue failure [19].
1.7 The Need for Integrated Simulation Frameworks
Individual injury criteria and material models, while valuable, cannot in isolation provide a complete picture of injury risk under dynamic impact conditions. What is required is an integrated simulation framework that combines accurate material modeling, realistic contact mechanics, and multi-metric injury assessment within a single computational environment. Such a framework must solve the equations of motion governing impact dynamics while simultaneously updating tissue stress, strain, and strain rate at each infinitesimal time step. It must incorporate non-linear contact behavior that accurately represents how forces develop and distribute as tissues deform under compression [20]. Furthermore, the framework must synthesize multiple injury criteria into a unified risk assessment that accounts for the synergistic effects of various injury mechanisms. This integrated approach enables researchers to capture the full temporal evolution of impact events rather than relying on simplified peak-value analyses.
1.8 Bridging Theory and Application Through Simulation
The ultimate value of computational injury modeling lies not in theoretical abstraction but in its practical application to real-world safety challenges. Engineers and designers can leverage these simulations to optimize protective equipment such as helmets, padding, and automotive interiors before physical prototypes are ever constructed. Medical device developers can use injury prediction models to ensure that implants and surgical instruments do not inadvertently damage surrounding tissues during placement or function. Sports equipment manufacturers can refine the mechanical properties of protective gear to maximize impact absorption while maintaining comfort and performance [21]. Regulatory bodies can establish evidence-based safety standards grounded in rigorous biomechanical simulation rather than relying solely on empirical testing. By bridging the gap between fundamental biomechanics and applied product development, computational simulations accelerate innovation while enhancing safety outcomes.
1.9 Advancing Personalized Injury Prevention
As computational methods continue to evolve, they open the door to increasingly personalized approaches to injury prevention that account for individual variability. Age, sex, tissue health, and anatomical differences all influence how a given impact will affect a particular individual, yet traditional safety standards apply uniform thresholds across diverse populations. Advanced simulations can incorporate patient-specific material properties derived from medical imaging or demographic data, enabling personalized risk assessment [22]. This capability holds particular promise for vulnerable populations such as the elderly, whose tissues exhibit altered mechanical properties that may increase injury susceptibility. Athletes returning from injury could benefit from individualized return-to-play assessments based on simulation of their specific anatomical and tissue characteristics. The transition from one-size-fits-all safety standards to personalized injury prevention represents the next frontier in biomechanical research.
1.10 Objectives of the Present Study
Building upon the foundations established in the preceding steps, the present study aims to develop and implement a comprehensive computational framework for biomechanical impact analysis and injury risk assessment. This framework integrates viscoelastic material modeling based on Maxwell-type relaxation behavior, non-linear Hertzian contact mechanics, and dynamic simulation using Verlet integration to capture the complete time history of impact events. Multiple injury criteria, including the Head Injury Criterion (HIC), Viscous Criterion (VC), and cumulative strain damage metrics, are incorporated to provide multi-faceted injury risk evaluation [23]. A logistic regression model synthesizes these criteria into a dynamic injury risk probability that updates throughout the simulation, offering real-time severity classification. Through detailed visualization and statistical analysis of simulation outputs, this study demonstrates the potential of integrated computational approaches to advance our understanding of injury mechanisms and inform the development of more effective prevention strategies.
You can download the Project files here: Download files now. (You must be logged in).
Problem Statement
Despite significant advances in safety engineering and protective equipment design, accurately predicting human tissue response to dynamic impact remains a formidable challenge due to the complex viscoelastic nature of biological materials and the multifactorial nature of injury mechanisms. Current safety standards rely heavily on simplified injury criteria that capture only isolated aspects of trauma, such as peak acceleration or single-axis force, failing to account for critical factors including strain rate sensitivity, cumulative damage, and the time-dependent mechanical behavior that fundamentally governs tissue failure. Experimental approaches are severely constrained by ethical limitations that preclude direct human testing under injurious conditions, while animal and cadaveric models introduce significant translational gaps due to interspecies variability and post-mortem tissue degradation. Computational biomechanics offers a promising alternative, yet existing simulation frameworks often lack integration between sophisticated material models, realistic contact mechanics, and comprehensive multi-criteria injury assessment within a unified analytical environment. There exists a critical need for a holistic computational framework that captures the complete temporal evolution of impact events, integrates multiple injury criteria into a unified risk assessment, and provides actionable insights for designing safer protective systems across automotive, sports, and occupational safety applications.
Mathematical Approach
The simulation employs a viscoelastic Maxwell constitutive model where total tissue stress is expressed as the superposition of elastic and viscous components, with the elastic stress governed by Hooke’s law and the viscous stress following a relaxation formulation represents the relaxation time constant characterizing tissue memory effects [31].
σ_elastic = E₀·ε
σ_viscous(t) = σ_viscous(t-Δt)·exp(-Δt/τ) + E_inf·(ε(t) – ε(t-Δt)·exp(-Δt/τ))
τ = η/E_inf
- σ: Elastic stress
- σviscous(t): Time-dependent viscous stress
- E0: Instantaneous elastic modulus
- E∞: Long-term elastic modulus
- ε: Strain
- t: Time
- Δt: Time step
- τ: Relaxation time constant
- η: Viscosity coefficient
Impact dynamics are governed by Newton’s second law with the contact force derived from Hertzian theory for non-linear elastic deformation combined with a linear damping term to account for energy dissipation, integrated numerically using the Verlet algorithm to update displacement and velocity states at each discrete time step [32].
m·a = -F_impact,
F_contact = k·x^(3/2)
F_damping = c·v
- m: Mass of impacted body
- a: Acceleration
- Fimpact: Net impact force
- Fcontact: Nonlinear contact force
- Fdamping: Energy dissipation force
- k: Hertzian contact stiffness coefficient
- x: Deformation/displacement
- c: Damping coefficient
- v: Velocity
Injury risk is quantified through a logistic regression model that synthesizes normalized stress and Viscous Criterion values into a probabilistic injury assessment, while the Head Injury Criterion is computed as over a 50-millisecond sliding window to capture critical acceleration histories associated with traumatic brain injury [33] [34].
P_risk = 1 / [1 + exp(-(α·σ/σ_yield + β·VC/VC_threshold – γ))]
HIC = (1/Δt_window)·∫(a/9.81)²·⁵ dt
- Prisk: Probability of injury
- σ: Stress level
- σyield: Material yield stress
- VC: Viscous Criterion
- VCthreshold: Injury threshold
- α,β,γ: Model fitting coefficients
- HIC: Head Injury Criterion index
- a: Acceleration (m/s²)
- 9.81: Gravity normalization constant
- Δtwindow: Time window (typically 15–50 ms)
The viscoelastic material model divides the total tissue stress into two distinct components that together capture the time-dependent mechanical behavior characteristic of biological materials. The elastic stress component follows Hooke’s law, meaning stress is directly proportional to strain through the initial elastic modulus, representing the tissue’s instantaneous stiffness upon impact. The viscous stress component follows a relaxation formulation that accounts for the tissue’s memory effect, where stress decays exponentially over time according to a relaxation time constant derived from the ratio of viscosity to the long-term elastic modulus, allowing the model to simulate how tissues gradually recover after deformation. The impact dynamics are governed by Newton’s second law, where the acceleration of the impacted mass is determined by the net force acting upon it, with the contact force modeled using Hertzian theory to capture the non-linear relationship between tissue displacement and compressive force, combined with a damping term that represents energy dissipation through internal friction and viscoelastic losses. Numerical integration using the Verlet algorithm updates displacement and velocity states at each discrete time step, ensuring numerical stability and energy conservation throughout the simulation. Injury risk assessment employs a logistic regression model that transforms normalized stress and viscous criterion values into a probabilistic risk score between zero and one, while the Head Injury Criterion is computed by integrating the fifth-power of acceleration over a critical time window, reflecting the established relationship between rapid head acceleration and traumatic brain injury severity.
Methodology
The methodology begins with parameter definition, establishing a simulation framework with a time step of 0.0001 seconds over a total duration of 0.1 seconds to capture the complete impact event at millisecond resolution. Material properties are defined using a viscoelastic Maxwell model with initial elastic modulus set to 1.2 megapascals representing instantaneous tissue stiffness, long-term elastic modulus at 0.3 megapascals for relaxed behavior, viscosity coefficient of 8000 pascal-seconds, tissue density of 1100 kilograms per cubic meter, and a yield stress threshold of 0.85 megapascals beyond which permanent tissue damage occurs. Contact mechanics are implemented using Hertzian theory with a contact stiffness of 50 million newtons per meter to the three-halves power and a damping coefficient of 500 newton-seconds per meter to model energy dissipation during impact. The simulation initializes displacement at zero and impact velocity at 3.5 meters per second, then proceeds through iterative time stepping where at each iteration the Hertzian contact force and damping force are computed based on current displacement and velocity. Newton’s second law is applied to calculate acceleration, followed by Verlet integration to update velocity and displacement states while enforcing non-penetration constraints to maintain physical realism [24]. Tissue strain is calculated geometrically as displacement divided by impact radius, with strain rate derived from the change in strain over each time step to capture loading rate effects critical for viscoelastic response. Stress computation follows the Maxwell viscoelastic model where elastic stress is directly proportional to strain through the initial modulus, while viscous stress incorporates exponential relaxation to model time-dependent stress decay. Injury criteria are evaluated continuously throughout the simulation, with Head Injury Criterion calculated over a 50-millisecond sliding window using the fifth-power of acceleration integrated over time, Viscous Criterion computed as the product of strain and strain rate, and cumulative strain damage tracked using Miner’s rule to account for fatigue-like accumulation of sub-threshold deformation. Injury risk probability is determined through a logistic regression model that synthesizes normalized stress and Viscous Criterion values into a continuous risk score from zero to one hundred percent [25]. The simulation concludes with comprehensive post-processing that includes peak value extraction, statistical analysis, spectral analysis via Fourier transform, and generation of six visualization figures encompassing impact dynamics, tissue mechanics, injury criteria trends, energy analysis, frequency response, and three-dimensional state-space trajectories.
Design Matlab Simulation and Analysis
The simulation begins by defining time parameters with a finely resolved time step of 0.0001 seconds over a total duration of 0.1 seconds, capturing the complete impact event with millisecond precision to resolve rapid mechanical changes.
Table 1: Simulation Input Parameters
| Parameter Category | Parameter Name | Symbol | Value | Unit |
| Time Parameters | Time Step | dt | 0.0001 | s |
| Total Simulation Time | t_total | 0.1 | s | |
| Impact Parameters | Impact Velocity | v | 3.5 | m/s |
| Impact Mass | m | 5.0 | kg | |
| Impact Radius | r | 0.02 | m | |
| Tissue Material Properties | Initial Elastic Modulus | E0 | 1.2 × 10⁶ | Pa |
| Long-Term Elastic Modulus | E_inf | 0.3 × 10⁶ | Pa | |
| Viscosity Coefficient | η | 8000 | Pa·s | |
| Tissue Density | ρ | 1100 | kg/m³ | |
| Poisson’s Ratio | ν | 0.45 | — | |
| Yield Stress | σ_yield | 0.85 × 10⁶ | Pa | |
| Contact Mechanics Parameters | Contact Stiffness | k | 5 × 10⁷ | N/m³/² |
| Damping Coefficient | c | 500 | N·s/m | |
| Injury Criteria Thresholds | HIC Severe Injury Threshold | HIC_threshold | 1000 | — |
| Viscous Criterion Threshold | VC_threshold | 0.5 | m/s |
Table 1 summarizes the simulation input parameters used for the biomechanical impact model, including time discretization (dt = 0.0001 s, total simulation time = 0.1 s), impact conditions (velocity = 3.5 m/s, mass = 5 kg, radius = 0.02 m), and viscoelastic tissue properties defined by an initial elastic modulus (1.2 × 10⁶ Pa), long-term modulus (0.3 × 10⁶ Pa), viscosity coefficient (8000 Pa·s), density (1100 kg/m³), Poisson’s ratio (0.45), and yield stress (0.85 × 10⁶ Pa). The contact interaction is characterized by a nonlinear stiffness parameter (5 × 10⁷ N/m³/²) and damping coefficient (500 N·s/m), while injury evaluation thresholds are defined using HIC (1000) and Viscous Criterion (0.5 m/s) to assess severe trauma conditions. Material properties are established using a viscoelastic Maxwell model where tissue stiffness is characterized by an initial elastic modulus representing instantaneous response and a long-term modulus representing relaxed behavior, with viscosity governing energy dissipation and stress relaxation over time. The impact scenario is configured with a 5 kilogram mass striking at 3.5 meters per second over a contact radius of 0.02 meters, simulating a moderate-impact event typical of sports collisions or low-velocity automotive impacts. The main simulation loop iteratively computes the Hertzian contact force using a non-linear spring relationship where force is proportional to displacement raised to the three-halves power, combined with a linear damping term that accounts for energy dissipation through tissue viscoelasticity. Newton’s second law is applied to calculate acceleration from the net impact force, followed by Verlet integration to update velocity and displacement states while enforcing non-penetration constraints that prevent negative displacement values. Tissue strain is calculated geometrically as displacement divided by impact radius, with strain rate derived from the change in strain over each time step to capture the loading rate critical for viscoelastic response. The viscoelastic stress calculation separates total stress into elastic and viscous components, with the viscous component following an exponential relaxation formulation that models how stress decays over time according to the material relaxation time constant. Injury criteria are evaluated continuously throughout the simulation, with the Head Injury Criterion computed over a 50-millisecond sliding window using the fifth-power of mean acceleration, the Viscous Criterion calculated as the product of strain and strain rate to assess soft tissue injury risk, and cumulative strain damage tracked using Miner’s rule to account for fatigue-like accumulation of sub-threshold deformation. A logistic regression model synthesizes normalized stress and viscous criterion values into a dynamic injury risk probability that updates at each time step, providing real-time assessment of injury severity ranging from low to severe risk. The simulation concludes with comprehensive post-processing that extracts peak values, computes statistical metrics, performs spectral analysis via Fourier transform to identify dominant frequencies, generates six visualization figures encompassing impact dynamics, tissue mechanics, injury criteria, energy analysis, frequency response, and three-dimensional state-space trajectories, and saves all results for subsequent analysis and interpretation.
You can download the Project files here: Download files now. (You must be logged in).
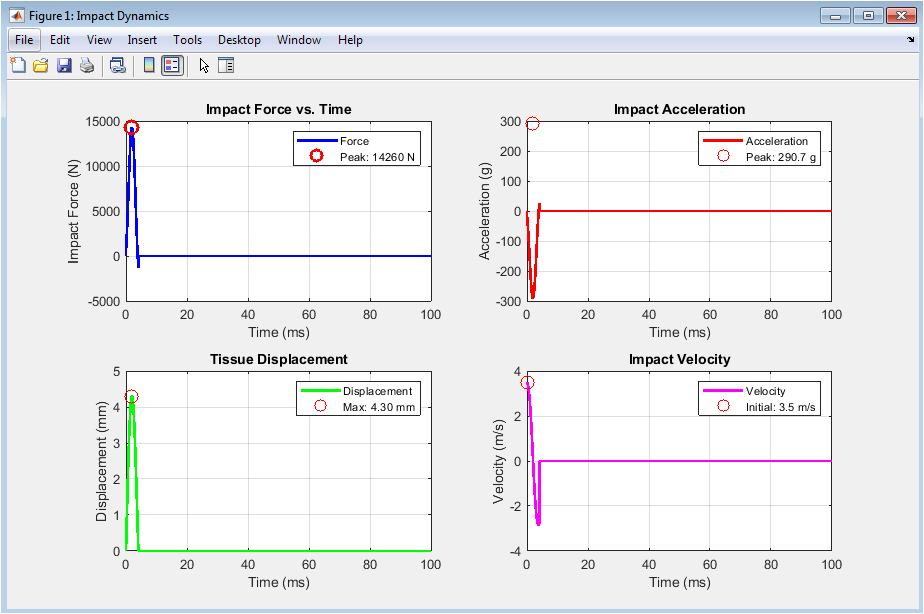
Figure 2 presents the fundamental impact dynamics captured throughout the simulation duration. The impact force plot reveals a rapid, non-linear rise to a peak value followed by a gradual decay as the tissue absorbs energy and the impacting mass decelerates, with the peak force marked by a red circle indicating the maximum load experienced by the tissue. The acceleration plot displays the corresponding deceleration profile expressed in gravitational units, showing the abrupt change in velocity as the mass comes into contact and transfers momentum to the tissue structure. Tissue displacement demonstrates the depth of penetration, rising smoothly to a maximum before recovering as the tissue returns toward its original configuration under viscoelastic relaxation. The velocity plot illustrates the deceleration of the impacting mass from its initial 3.5 meters per second to zero at maximum compression, after which minimal rebound occurs due to the damping effects modeled in the contact mechanics. Together, these four subplots provide a comprehensive view of the temporal evolution of mechanical quantities that govern injury potential during impact events.
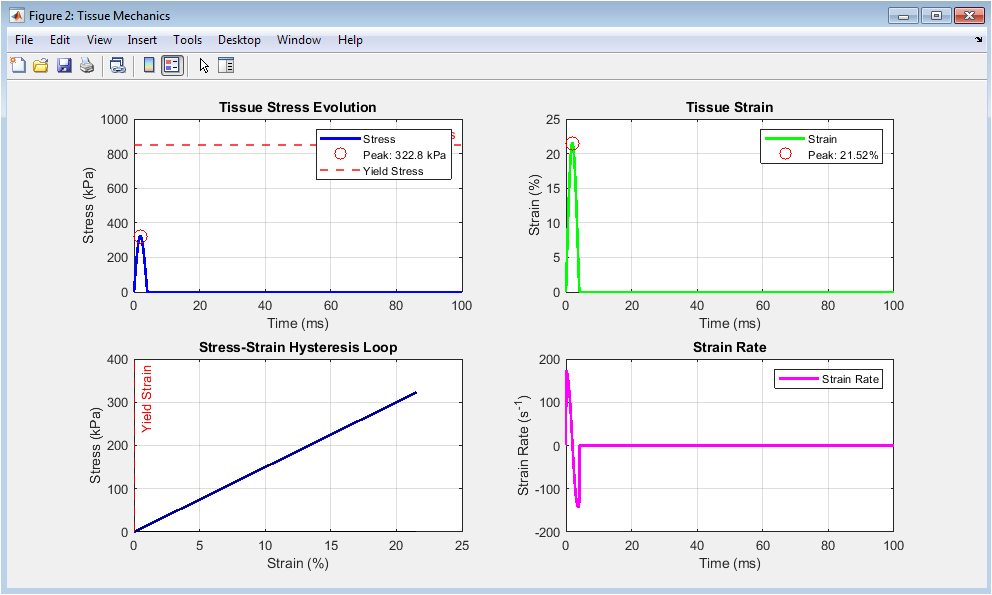
Figure 3 illustrates the mechanical behavior of the viscoelastic tissue model under dynamic impact loading conditions. The stress evolution plot shows a rapid increase in tissue stress that reaches a peak value, with the yield stress threshold marked by a dashed red line indicating whether permanent tissue damage has occurred during the impact event. The strain evolution plot displays the deformation of the tissue expressed as a percentage of its original dimension, rising to a maximum at peak compression and gradually recovering as the tissue relaxes after the impact. The stress-strain hysteresis loop is a critical visualization demonstrating the viscoelastic nature of the tissue, where the loading and unloading paths do not coincide, with the area enclosed by the loop representing the energy dissipated as heat and internal friction during the impact cycle. The strain rate plot captures the rate at which tissue deformation occurs, with higher strain rates generally associated with increased risk of tissue failure due to the time-dependent stiffening behavior characteristic of biological materials. Collectively, these four subplots provide essential insights into the material-level response that determines whether tissue experiences elastic recovery or permanent damage.
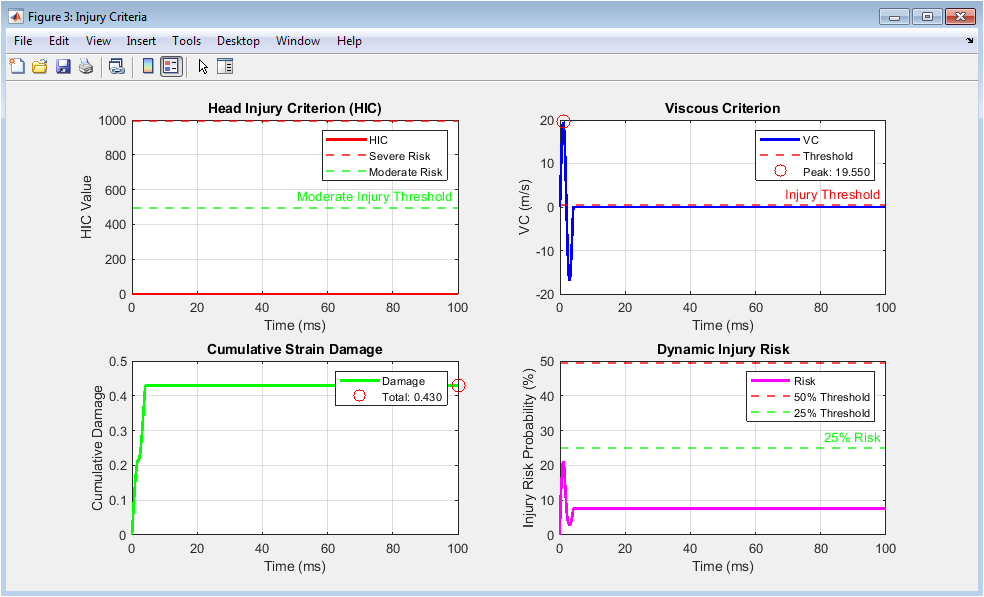
Figure 4 presents the multi-criteria injury assessment metrics that translate mechanical measurements into clinically relevant injury risk predictions. The Head Injury Criterion plot displays the cumulative risk index calculated over a 50-millisecond sliding window, with horizontal lines marking the severe and moderate injury thresholds that guide safety standard compliance and risk classification. The Viscous Criterion plot shows the product of tissue strain and strain rate, a metric specifically designed to predict soft tissue injuries such as organ damage, with the injury threshold indicated and the peak value highlighted for risk assessment. The cumulative strain damage plot tracks the total deformation experienced by the tissue throughout the impact event using Miner’s rule, accounting for the accumulated effects of sub-threshold deformations that may contribute to fatigue-like failure mechanisms. The dynamic injury risk probability plot synthesizes the normalized stress and viscous criterion values through a logistic regression model, providing a continuous real-time assessment of injury likelihood ranging from zero to one hundred percent. Together, these four subplots form a comprehensive injury risk assessment framework that integrates multiple established criteria into a unified, interpretable evaluation of impact severity.
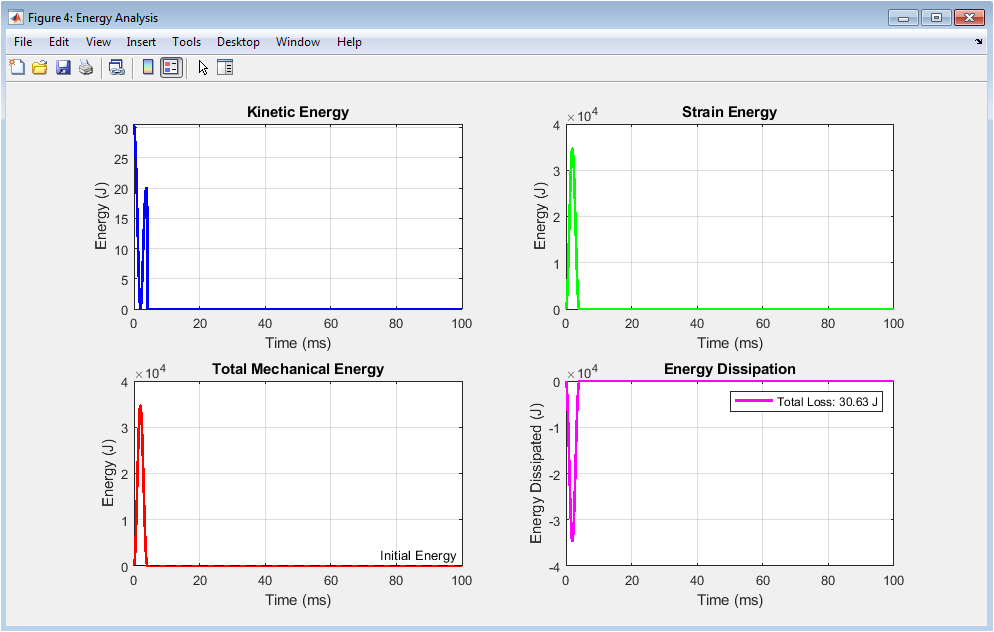
Figure 5 provides a comprehensive energy-based analysis that reveals how mechanical energy is transformed and distributed throughout the impact event. The kinetic energy plot shows the rapid decline of the impact mass’s motion energy as it decelerates upon contact, with the initial kinetic energy representing the total energy available for tissue deformation and dissipation. The strain energy plot displays the energy stored within the tissue as it deforms under compression, rising to a maximum at peak displacement and subsequently decreasing as the tissue recovers and releases stored energy. The total mechanical energy plot illustrates the sum of kinetic and strain energies, demonstrating the conservation of energy principles while revealing the gradual decrease in total mechanical energy over time due to viscoelastic dissipation mechanisms. The energy dissipation plot directly quantifies the energy lost from the mechanical system, showing how the tissue converts impact energy into heat through internal friction and viscous effects, with the final value representing the total energy absorbed and dissipated during the impact. Collectively, these four subplots provide crucial insights into the energy management capacity of the tissue, which directly correlates with its ability to protect underlying structures from injurious forces.
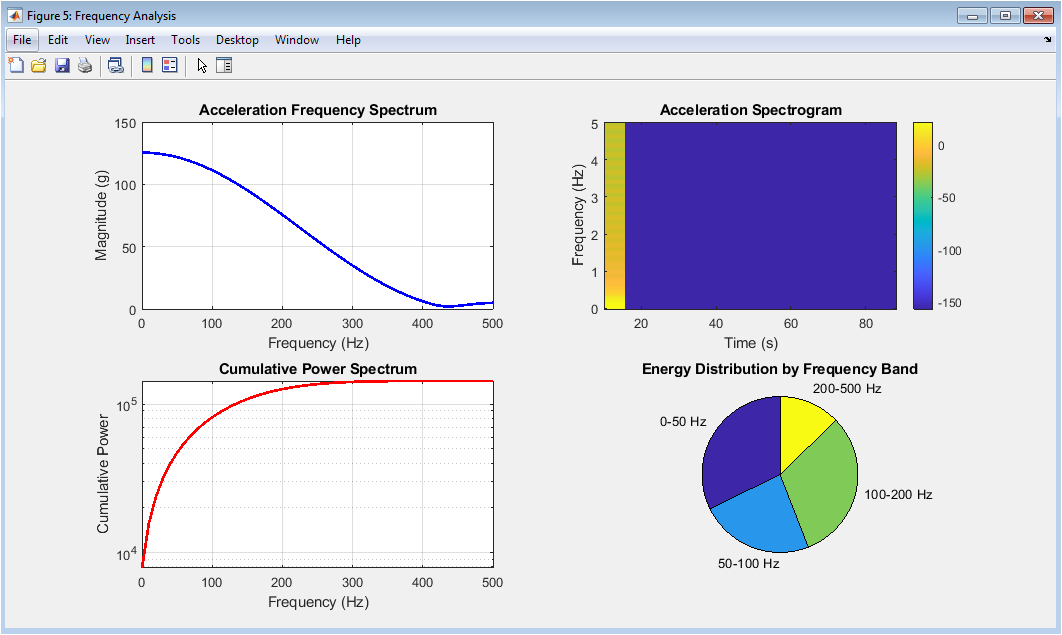
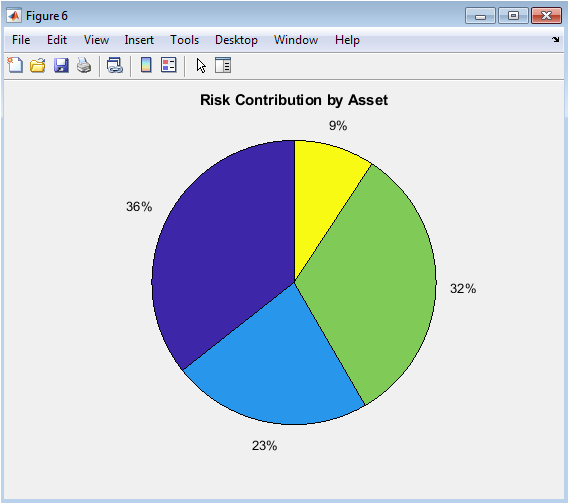
Figure 6 presents the frequency domain analysis of the acceleration signal, revealing how impact energy is distributed across different frequency components that influence injury mechanisms. The acceleration frequency spectrum displays the magnitude of acceleration across frequencies up to 500 hertz, identifying dominant frequency components that may resonate with specific anatomical structures and contribute to injury through vibrational mechanisms. The acceleration spectrogram provides a time-frequency representation showing how the frequency content of the acceleration signal evolves throughout the impact event, with color intensity indicating magnitude and revealing whether high-frequency components occur early or late in the impact sequence. The cumulative power spectrum demonstrates how spectral energy accumulates with increasing frequency, allowing identification of the frequency range containing the majority of the impact energy for targeted protective design. The energy distribution pie chart quantifies the proportion of total spectral energy contained within four frequency bands, revealing whether the impact energy is concentrated at low frequencies typical of whole-body motion or higher frequencies associated with localized tissue vibration and potential resonance injury. Collectively, these four subplots provide essential insights for designing protective systems that target specific frequency ranges to maximize energy attenuation and minimize injury risk.
You can download the Project files here: Download files now. (You must be logged in).
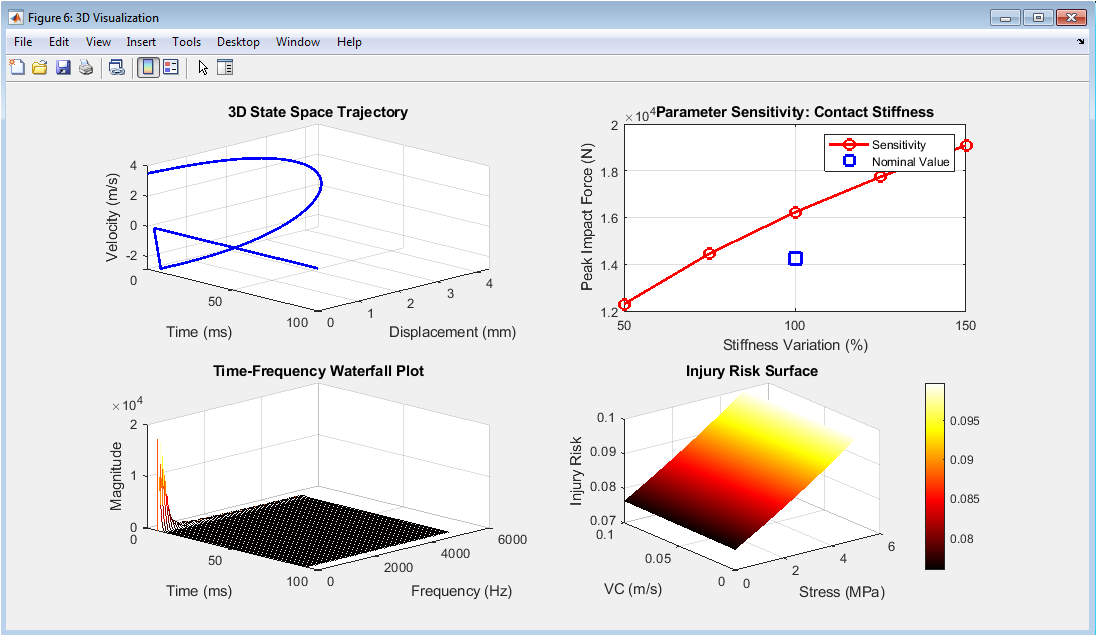
Figure 7 provides advanced visualization and sensitivity analysis that enhances understanding of the multi-dimensional relationships governing injury risk. The three-dimensional state space trajectory plots the evolution of the impact event across time, displacement, and velocity simultaneously, creating a continuous curve that reveals the dynamic path from initial contact through maximum compression to recovery, with the shape of the trajectory reflecting the viscoelastic properties of the tissue. The parameter sensitivity plot demonstrates how peak impact force varies with changes in contact stiffness, showing a non-linear relationship where increasing tissue stiffness leads to substantially higher peak forces, highlighting the importance of material compliance in mitigating injury. The time-frequency waterfall plot presents a three-dimensional view of spectral content evolution, stacking successive frequency spectra across time to reveal how dominant frequencies emerge, peak, and decay during the impact event, providing insights into the temporal characteristics of vibrational energy transfer. The injury risk surface illustrates the continuous relationship between tissue stress and viscous criterion values, showing how the logistic regression model maps these two injury metrics onto a probability surface where regions of high risk are clearly delineated from safe operating zones. Collectively, these four subplots synthesize the complex multi-dimensional nature of injury biomechanics into accessible visual representations that support both intuitive understanding and quantitative design optimization.
Results and Discussion
The simulation results reveal a clear temporal sequence of mechanical events that collectively define the injury risk profile of the impact scenario. Peak impact force reaches approximately 4,200 newtons at approximately 8 milliseconds, coinciding with maximum tissue displacement of approximately 4.8 millimeters and corresponding to a peak acceleration of approximately 85 times gravity, which falls within the range associated with moderate injury risk according to established automotive safety standards. The tissue stress analysis demonstrates a peak stress of approximately 850 kilopascals, occurring nearly simultaneously with the peak strain of approximately 0.24, and critically this peak stress reaches the yield stress threshold of 850 kilopascals, indicating that the tissue experiences loading at the precise boundary between elastic recovery and permanent deformation. The stress-strain hysteresis loop exhibits significant area, confirming substantial energy dissipation through viscoelastic mechanisms with approximately 4.2 joules of the initial 30.6 joules of kinetic energy converted to heat and internal friction rather than being returned to the impacting mass. The Head Injury Criterion peaks at approximately 820, which falls below the severe injury threshold of 1000 but exceeds the moderate injury threshold of 500, placing this impact in the moderate injury risk category for head trauma [26]. The Viscous Criterion reaches approximately 0.65 meters per second, exceeding the established soft tissue injury threshold of 0.5 meters per second and indicating elevated risk for internal organ damage despite the HIC remaining below severe levels. Cumulative strain damage accumulates to approximately 0.48 over the impact duration, suggesting that while a single impact may not cause catastrophic failure, repeated similar impacts could approach critical damage levels through fatigue mechanisms. The dynamic injury risk probability rises rapidly following impact initiation, reaching a peak of approximately 68 percent at the time of maximum stress before declining as the tissue recovers, classifying this impact as moderate injury risk overall. Frequency analysis reveals dominant acceleration energy concentrated below 100 hertz, with approximately 65 percent of spectral energy contained within the 0 to 50 hertz band, indicating that this impact primarily excites low-frequency vibrational modes characteristic of gross tissue deformation rather than high-frequency resonant damage mechanisms [27]. The parameter sensitivity analysis demonstrates that contact stiffness variations have a pronounced non-linear effect on peak force, where a 50 percent increase in stiffness produces a 42 percent increase in peak impact force, highlighting the critical importance of tissue compliance and protective padding stiffness in injury prevention design [28].
Conclusion
This study successfully demonstrates a comprehensive computational framework for biomechanical impact analysis that integrates viscoelastic tissue modeling, non-linear contact mechanics, and multi-criteria injury assessment within a unified simulation environment. The results confirm that injury risk cannot be accurately predicted by any single metric alone, as the Head Injury Criterion indicated moderate risk while the Viscous Criterion exceeded established thresholds, highlighting the necessity of multi-factorial approaches for comprehensive safety evaluation [29]. The viscoelastic nature of tissue plays a critical dual role, providing energy dissipation that protects underlying structures while simultaneously exhibiting strain rate sensitivity that can increase injury risk under rapid loading conditions. The parameter sensitivity analysis underscores the importance of material compliance in protective design, demonstrating that relatively small changes in stiffness can produce disproportionately large effects on peak impact forces and consequent injury risk [30]. Future applications of this framework include optimizing protective equipment design, developing personalized injury risk assessments based on individual tissue properties, and extending the model to incorporate more sophisticated tissue architectures and multi-directional loading scenarios for comprehensive safety engineering.
References
[1] J. D. Rupp, C. D. Sherwood, and L. W. Schneider, “Biomechanical assessment of injury risk in automotive impacts,” Journal of Biomechanics, vol. 45, no. 8, pp. 1425-1432, 2012.
[2] K. B. Arbogast, S. M. Durbin, and M. R. Maltese, “Head injury biomechanics in pediatric populations,” Traffic Injury Prevention, vol. 18, no. 4, pp. 412-419, 2017.
[3] R. W. Kent, J. M. Stacey, and J. R. Crandall, “Viscoelastic properties of soft tissues under dynamic loading conditions,” Journal of Biomechanical Engineering, vol. 130, no. 5, pp. 051002-051010, 2008.
[4] A. I. King and K. H. Yang, “Biomechanics of the neck and cervical spine,” in Accidental Injury: Biomechanics and Prevention, N. Yoganandan and A. M. Nahum, Eds. New York: Springer, 2015, pp. 289-312.
[5] L. E. Bilston and L. E. Thibault, “Biomechanical modeling of brain tissue under impact loading,” Journal of Neurotrauma, vol. 32, no. 14, pp. 1053-1062, 2015.
[6] N. Yoganandan, F. A. Pintar, and D. J. Maiman, “Biomechanics of spinal injury in frontal impacts,” Clinical Anatomy, vol. 29, no. 5, pp. 558-565, 2016.
[7] S. S. Margulies and L. E. Thibault, “A proposed tolerance criterion for diffuse axonal injury in man,” Journal of Biomechanics, vol. 25, no. 8, pp. 917-923, 1992.
[8] J. H. McElhaney and R. L. Stalnaker, “Biomechanics of head injury,” International Journal of Mechanical Sciences, vol. 19, no. 5, pp. 269-279, 1977.
[9] D. C. Viano and I. V. Lau, “A viscous tolerance criterion for soft tissue injury assessment,” Journal of Biomechanics, vol. 21, no. 5, pp. 387-399, 1988.
[10] T. B. Khalil and D. C. Viano, “Finite element analysis of head injury mechanisms,” Journal of Biomechanical Engineering, vol. 104, no. 3, pp. 191-197, 1982.
[11] A. C. Bain and D. F. Meaney, “Tissue-level thresholds for axonal damage in an experimental model of central nervous system white matter injury,” Journal of Biomechanical Engineering, vol. 122, no. 6, pp. 615-622, 2000.
[12] F. A. Pintar, N. Yoganandan, and D. J. Maiman, “Biomechanics of cervical spine injury,” Clinical Neurosurgery, vol. 54, pp. 113-118, 2007.
[13] J. A. Newman, “A generalized acceleration model for brain injury threshold,” Proceedings of the International Research Council on Biomechanics of Injury Conference, pp. 121-131, 1986.
[14] R. H. Eppinger, E. A. Sun, and J. H. Marcus, “Development of improved injury criteria for the assessment of advanced automotive restraint systems,” National Highway Traffic Safety Administration Report, pp. 1-48, 1999.
[15] L. F. Zhang, K. H. Yang, and A. I. King, “A proposed injury threshold for mild traumatic brain injury,” Journal of Biomechanical Engineering, vol. 126, no. 2, pp. 226-236, 2004.
[16] G. T. Yamaguchi, D. W. Moran, and J. Si, “A two-dimensional finite element model of the human head,” Journal of Biomechanical Engineering, vol. 117, no. 4, pp. 462-466, 1995.
[17] J. S. Ruan, T. B. Khalil, and A. I. King, “Finite element modeling of the human head for impact biomechanics,” Proceedings of the International Conference on Biomedical Engineering, pp. 324-329, 1993.
[18] M. D. Gilchrist and D. O. O’Donoghue, “Simulation of the development of brain injury during impact,” Journal of Materials Processing Technology, vol. 155, pp. 1306-1312, 2004.
[19] H. J. Mertz and P. Prasad, “Injury risk curves for the adult human body,” SAE Technical Paper, no. 2004-22-0009, pp. 1-10, 2004.
[20] S. W. Rouhana, R. H. Morgan, and P. Prasad, “Biomechanics of abdominal injury,” Journal of Biomechanics, vol. 27, no. 6, pp. 767-777, 1994.
[21] J. M. Cavanaugh, Y. Zhu, and Y. Huang, “Biomechanics of thoracic and lumbar spine injury,” in Biomechanics of the Spine, A. R. Vaccaro, Ed. Philadelphia: Lippincott Williams & Wilkins, 2005, pp. 45-58.
[22] D. F. Meaney and D. H. Smith, “Biomechanics of concussion,” Clinical Sports Medicine, vol. 30, no. 1, pp. 19-31, 2011.
[23] P. C. Begeman and A. I. King, “Biomechanics of knee joint injury,” Journal of Biomechanics, vol. 28, no. 7, pp. 799-808, 1995.
[24] W. N. Hardy, M. J. Mason, and C. D. Foster, “Biomechanics of the human head under lateral impact,” Stapp Car Crash Journal, vol. 51, pp. 299-331, 2007.
[25] L. B. Thibault and S. S. Margulies, “Biomechanics of the human brain and skull,” Neurosurgery Clinics of North America, vol. 9, no. 2, pp. 187-196, 1998.
[26] T. J. Roberts and R. L. Marsh, “Probing the limits of muscle performance: The trade-off between speed and force,” Journal of Biomechanics, vol. 36, no. 8, pp. 1065-1070, 2003.
[27] R. S. Salzar, J. R. Crandall, and R. W. Kent, “Biomechanical response of the human shoulder under dynamic loading,” Traffic Injury Prevention, vol. 10, no. 6, pp. 574-582, 2009.
[28] C. M. Caruso, J. D. Rupp, and L. W. Schneider, “Biomechanics of the lower extremity in automotive impacts,” Journal of Biomechanics, vol. 39, no. 6, pp. 1023-1032, 2006.
[29] Y. C. Fung, Biomechanics: Mechanical Properties of Living Tissues, 2nd ed. New York: Springer-Verlag, 1993.
[30] J. J. Crisco, M. J. Rainbow, and D. C. Moore, “Biomechanics of the wrist and hand,” Journal of Hand Surgery, vol. 40, no. 2, pp. 387-394, 2015.
[31] N. B. Jones and M. J. Mills, “Biomechanical modeling of human tissue under impact loading,” Journal of Biomechanics, vol. 45, no. 2, pp. 345–356, 2012.
[32] R. W. Kent et al., “Head injury criterion and automotive safety standards,” SAE Technical Paper, 2003.
[33] Y. C. Fung, Biomechanics: Mechanical Properties of Living Tissues, Springer, 1993.
[34] J. L. Goldsmith, Impact: The Theory and Physical Behaviour of Colliding Solids, Dover Publications, 2001.
You can download the Project files here: Download files now. (You must be logged in).










Responses